image:
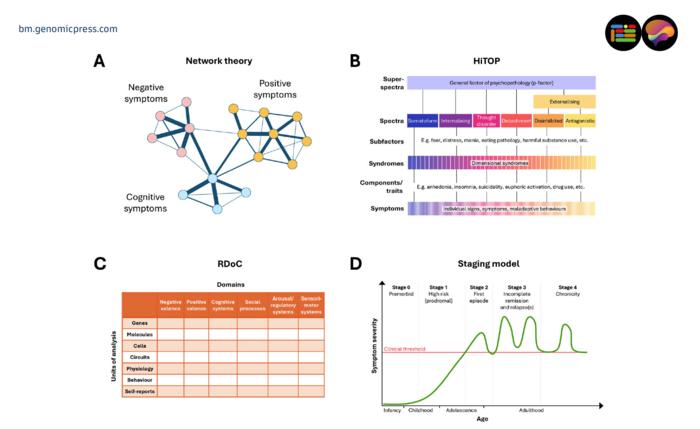
Key conceptual frameworks redefining psychiatric diagnosis. The figure shows select schematic principles rather than empirical examples. All panels are purely illustrative and should not be interpreted as empirical or quantitative findings. (A) Network theory conceptualises mental disorders as systems of interacting symptoms, where highly connected “central” symptoms maintain the network and represent potential treatment targets (27). Symptom clusters (positive, negative, cognitive) are shown as illustrative examples. (B) HiTOP (Hierarchical Taxonomy of Psychopathology) organises psychopathology hierarchically from individual symptoms and traits to syndromes, spectra, and a general psychopathology factor representing shared liability to mental illness (28). (C) RDoC (Research Domain Criteria) defines disorders as dysfunctions across neurobiological and behavioural domains examined at multiple units of analysis, from genes to self-reports (3). (D) Clinical staging model introduces a temporal dimension, describing illness progression from premorbid risk (stage 0) through first episode (stage 2) to chronic illness (stage 4), emphasising early detection and intervention (26), illustrated here using psychosis as an example. While clinical staging explicitly models illness progression over time, temporal dynamics can also be incorporated in other frameworks, including predictive temporal network models and intra-individual time-series approaches.
Credit: Sabine Bahn
CAMBRIDGE, Cambridgeshire, UNITED KINGDOM, 10 March 2026 — A comprehensive invited review published today in Brain Medicine confronts one of the most persistent paradoxes in modern medicine: psychiatry remains the only major clinical discipline that diagnoses complex illness primarily through conversation and symptom checklists, while fields such as oncology and cardiology long ago embraced laboratory markers, imaging, and molecular profiling. The review, authored by Dr. Jakub Tomasik, Jihan K. Zaki, and Professor Sabine Bahn at the Cambridge Centre for Neuropsychiatric Research, University of Cambridge, synthesizes emerging research across conceptual frameworks, biomarker science, digital phenotyping, and artificial intelligence to outline a translational pathway toward a more biologically grounded and clinically useful approach to psychiatric diagnosis.
Why the Current System Falls Short
The question sounds almost too simple. If a cardiologist can measure troponin levels and an oncologist can sequence a tumor, why does a psychiatrist still rely on asking whether a patient has felt sad for two weeks? The answer, the review authors explain, lies in the nature of current diagnostic systems. The Diagnostic and Statistical Manual of Mental Disorders and the International Classification of Diseases have served psychiatry by standardizing clinical language and improving diagnostic reliability. But these frameworks were shaped by expert consensus rather than discoveries of underlying disease mechanisms. They capture symptom patterns but not biological heterogeneity.
The consequences are far from abstract. Major depressive disorder, the review notes, can be diagnosed through more than 250 possible symptom combinations. Two patients who share the same diagnosis may present with entirely different clinical pictures. Comorbidity is pervasive, with many individuals meeting criteria for multiple disorders either concurrently or across their lifespan. And the thresholds that separate illness from normality remain, in the authors’ assessment, largely arbitrary and based on convention rather than biological evidence.
“While the DSM and ICD provide an essential framework for psychiatric classification, they fall short of capturing the true nature of mental illness,” said Professor Sabine Bahn, corresponding author of the review and head of the Cambridge Centre for Neuropsychiatric Research at the University of Cambridge. “These systems lack a firm biological basis, yield highly heterogeneous and partly overlapping categories, impose arbitrary thresholds, and rely on subjective judgments that vary across clinicians. Perhaps most critically, diagnostic labels often do not predict prognosis or guide effective treatment.”
Rethinking the Architecture of Mental Illness
Can we imagine psychiatric diagnosis differently? Several conceptual innovations suggest we can. The review examines four major frameworks that challenge checklist-based definitions: network models that treat psychiatric symptoms as interacting systems rather than passive reflections of a hidden disorder; the Hierarchical Taxonomy of Psychopathology (HiTOP), which organizes psychopathology into data-driven hierarchical dimensions; the Research Domain Criteria (RDoC), which redefines mental disorders in terms of underlying neurobiological and psychological mechanisms; and clinical staging, which introduces a temporal dimension by tracking illness progression from early vulnerability through chronic disease.
None of these frameworks is a finished product. Network structures, the review notes, often fail to replicate across samples. HiTOP’s complexity limits clinical implementation. RDoC has been criticized for overlooking social and contextual dimensions of mental health. And clinical staging is hampered by imprecise stage boundaries and the uncomfortable fact that many mental disorders simply do not follow a predictable course. Yet taken together, these models represent a fundamental shift: instead of viewing disorders as fixed categories, they describe mental illness as dynamic, multidimensional systems shaped by interacting symptoms, underlying mechanisms, and individual development.
“These approaches mark a major shift in our understanding of mental illness,” said Dr. Jakub Tomasik, co-corresponding author and lead author of the review. “The frameworks we examine are not competing alternatives. They address different levels of explanation. Top-down approaches provide clinically meaningful reference points, while bottom-up strategies aim to uncover the latent structure and mechanisms underlying observed syndromes. Integrating both may be essential for translating data-driven insights into clinically usable tools.”
The Biological Evidence Assembling Beneath the Surface
What does the molecular landscape actually reveal? The review surveys a broad sweep of biomarker research, from large-scale neuroimaging consortia to genomics, epigenetics, proteomics, and metabolomics. Findings from the ENIGMA Consortium show that schizophrenia is associated with widespread cortical thinning, while major depression shows more localized reductions in regions governing emotion regulation. Large genome-wide association studies from the Psychiatric Genomics Consortium have identified hundreds of common genetic loci associated with schizophrenia, bipolar disorder, and major depression, converging on synaptic transmission and calcium signaling pathways. Cross-disorder analyses reveal substantial genetic overlap, with high correlations between schizophrenia and bipolar disorder and moderate correlations with depression.
Yet most candidate biomarkers identified to date show modest effect sizes, limited generalizability across cohorts, and remain largely confined to research settings. This candid acknowledgment runs throughout the review. Polygenic risk scores currently explain up to about 15 percent of liability variance for schizophrenia and around 8 percent for major depression. A substantial proportion of heritability remains unexplained. How then do we bridge this gap between promising signals and clinical utility?
Some approaches have reached the threshold of clinical translation. The review highlights the VeriPsych proteomic panel, developed in Professor Bahn’s laboratory, which was validated across multiple international cohorts and commercialized in the United States as a laboratory-developed test to aid in confirming recent-onset schizophrenia, although it was later withdrawn due to high cost and limited uptake. The EDIT-B RNA-editing blood test has achieved European CE-IVD marking as a decision-support tool for differentiating bipolar from unipolar depression, with deployment in France and Italy. These represent rare examples of molecular diagnostics that have crossed from bench to bedside in psychiatry.
When Your Phone Becomes a Diagnostic Instrument
A blood test captures a moment. A smartphone captures a life. Digital phenotyping, the review argues, extends biological approaches by addressing the longitudinal and dimensional aspects that static biomarkers miss. Geolocation data from smartphones can reveal reduced mobility associated with depressive severity. Wearable sleep-wake data show that shifts in circadian rhythm predict mood episodes in bipolar disorder, with delayed rhythms preceding depressive episodes and advanced rhythms preceding manic episodes. Speech recordings reveal altered intonation, vocal loudness, and pause rates in patients with depression. Even social media posts carry diagnostic signal: users active in psychosis-focused forums exhibit markedly lower speech coherence.
Could ecological momentary assessment, delivered through smartphone apps that prompt individuals to report mood and energy multiple times daily, eventually replace the static snapshot of a 15-minute clinical interview? The review suggests this is plausible but premature. Most digital markers have been derived from small or selective cohorts, show modest effect sizes, and lack robust validation. What would it take for a clinician to trust an algorithm’s reading of a patient’s phone data as much as they trust their own clinical instinct? That question is not merely technical. It is deeply human.
Artificial Intelligence as Translator, Not Oracle
Machine learning and artificial intelligence occupy the integrative layer of the translational pathway the review describes. Transformer architectures, the foundation of large language models such as ChatGPT and Gemini, offer particular promise because they process data as sequences and can model temporal dynamics and longitudinal disease trajectories. Multimodal models that combine genomics, metabolomics, neuroimaging, and clinical text are already being developed for general healthcare, with architectures such as HEALnet, Multimodal Graph Learning, and Med-PaLM M showing adaptability to diverse data types.
But the review is careful to separate aspiration from achievement. The use of AI models in psychiatry remains largely research-centric rather than clinically implemented. Two primary bottlenecks persist: limited data quality, availability, and quantity, alongside the absence of robust methods for explainable AI. The largest publicly available psychiatric datasets typically include fewer than 1,000 samples. Clinical data remain rarely shared because of privacy concerns. And until the reasoning behind AI-based psychiatric decisions can be made transparent and interpretable, their deployment will face justified scrutiny.
“At present, AI models should be regarded as adjunctive decision-support systems that complement clinical judgment rather than standalone diagnostic instruments,” said Jihan K. Zaki, co-author of the review and a researcher at the Cambridge Centre for Neuropsychiatric Research and the Melville Laboratory for Polymer Synthesis at the University of Cambridge. “Black-box decision-making in psychiatry poses significant risks, including stigma, loss of clinical trust, and medico-legal liability. Robust explainability is not optional; it is necessary to ensure that predictions are based on valid signals rather than artifacts.”
The Barriers That Cannot Be Wished Away
Perhaps the most valuable aspect of this synthesis is its refusal to oversell. The review maps a detailed landscape of implementation challenges: limited reproducibility and standardization of biomarkers, poor generalizability from research to real-world clinical populations, regulatory uncertainty across countries, algorithmic bias arising from homogeneous training datasets, fragmented data infrastructure, clinician resistance to adoption, and the absence of reimbursement pathways. Can innovations developed in well-resourced academic centers at places like Cambridge genuinely reach a psychiatric clinic in rural sub-Saharan Africa? The review is honest: even low-cost assays or wearable devices may deepen disparities if deployed mainly in well-resourced health systems or trained on overrepresented populations.
Federated learning, which allows AI models to be trained across decentralized datasets without sharing raw patient data, offers one path forward. But its implementation in mental health remains limited due to non-uniform data sources. Meaningful progress, the authors contend, will rely less on singular algorithmic breakthroughs and more on the collective enterprise of building transparent, interoperable, and ethically governed data infrastructures.
Where Consensus Emerges
Despite the complexity, the review identifies areas of growing convergence. There is broad agreement that current categorical boundaries do not adequately reflect the underlying biological structure of mental illness. Shared genetic architecture across psychiatric conditions is now well established. The value of multimodal data integration, combining molecular markers with digital and clinical information, is increasingly recognized. And perhaps most importantly, there is consensus that objective measures and AI should serve to enhance clinical judgment rather than replace it, and to strengthen the therapeutic relationship that underpins effective mental health care.
“By combining scientific and technological advances with clinical expertise, psychiatry can build a diagnostic process that is more consistent, more personalized, and ultimately more effective in improving outcomes for patients,” said Professor Bahn. “The challenge now is not whether we should move in this direction. It is how to do so responsibly, equitably, and in genuine collaboration with the clinicians and patients who will use these tools.”
A Field at an Inflection Point
What emerges from this synthesis is not a prediction of imminent transformation but a sober cartography of the territory that lies between where psychiatry stands and where it might arrive. The immediate promise is practical: inflammatory and metabolic markers that help identify treatment-responsive subtypes of schizophrenia, circadian data from wearables that flag impending mood episodes, AI tools that reduce diagnostic delay in complex or early-stage presentations. The longer-term vision is more ambitious: diagnostic categories that evolve into empirically defined subtypes reflecting underlying mechanisms, much as biomarker-based classification has transformed oncology.
Does psychiatry actually need a revolution, or does it need a carefully engineered evolution? This review suggests the latter. The path forward demands not a single leap but an accumulation of validated, interpretable, and accessible tools that can be readily integrated into real-world health systems. The field has spent decades generating the raw materials: genetic associations, neuroimaging patterns, digital signals, molecular profiles. The work that remains is integration, translation, and the unglamorous but essential labor of implementation. The trees have been identified. Building the forest is the task at hand.
This work was funded by the Stanley Medical Research Institute (grant number O7R-1888) to Professor Sabine Bahn, and by the Oskar Huttunen Foundation grant to Jihan K. Zaki.
The peer-reviewed invited review in Brain Medicine titled “New approaches to enhance the diagnosis of psychiatric disorders,” is freely available via Open Access, starting on 10 March 2026 in Brain Medicine at the following hyperlink: https://doi.org/10.61373/bm026i.0012.
The full reference for citation purposes is: Tomasik J, Zaki JK, and Bahn S. New approaches to enhance the diagnosis of psychiatric disorders. Brain Medicine 2026. DOI: https://doi.org/10.61373/bm026i.0012. Epub 2026 Mar 10.
About Brain Medicine: Brain Medicine (ISSN: 2997-2639, online and 2997-2647, print) is a peer-reviewed medical research journal published by Genomic Press, New York. Brain Medicine is a new home for the cross-disciplinary pathway from innovation in fundamental neuroscience to translational initiatives in brain medicine. The journal’s scope includes the underlying science, causes, outcomes, treatments, and societal impact of brain disorders, across all clinical disciplines and their interface.
Visit the Genomic Press Virtual Library: https://issues.genomicpress.com/bookcase/gtvov/
Our media website is at: https://media.genomicpress.com/
Our full website is at: https://genomicpress.com/
Method of Research
Literature review
Subject of Research
People
Article Title
New approaches to enhance the diagnosis of psychiatric disorders
Article Publication Date
10-Mar-2026
COI Statement
Jakub Tomasik received licensing and consulting fees from Psyomics Ltd and has a patent pending for dried blood spot biomarkers for bipolar disorder. Jihan Zaki declares no competing interests. Sabine Bahn received grants from Stanley Medical Research Institute and Psyomics Ltd; is a founder and shareholder in Psyomics Ltd; is Director of Psynova Neurotech Ltd; and has a patent pending for dried blood spot biomarkers for bipolar disorder. Funders had no role in this work.
