The ‘wrong drug’ is in some boxes of madication taken by 2m Brits – with possible serious side effects if they are mixed
More than two million Britons take sertraline to help manage the symptoms of depression, anxiety and mood disorders(Image: Getty Images)
Health officials have released details of side effects to look out for after an urgent recall was issued for one of the most widely used antidepressants in the UK. The Medicines and Healthcare products Regulatory Agency (MHRA) has urged people to check their sertraline immediately.
Due to a manufacturing error, some packets of sertraline were found to contain a different medication entirely — and taking both could leave patients seriously ill. More than two million Britons rely on sertraline to manage symptoms of depression, anxiety and other mood disorders such as obsessive compulsive disorder (OCD).
The urgent recall was triggered after a patient discovered the wrong medicine inside their sealed carton, the MHRA confirmed. The patient found that a pack of Sertraline 100mg film-coated tablets contained one blister strip of Citalopram 40mg film-coated tablets, which is another antidepressant.
The ‘rogue’ tablets turning up in some patients’ boxes alongside a strip of sertraline 100mg are citalopram 40mg, a drug also commonly prescribed for mental health conditions. Both medications are classified as selective serotonin reuptake inhibitors (SSRIs), which work by boosting levels of the ‘happy hormone’ serotonin in the brain.

Sertraline 100 mg film-coated tablets – Batch Number V2500425, Outer carton and Lot (Batch Number) /EXP Date information.(Image: MHRA)
However, taking two different SSRIs together can trigger a dangerous condition known as serotonin syndrome, which can prove fatal. Equally, abruptly stopping SSRIs can bring on a range of debilitating withdrawal effects. Dr Alison Cave, MHRA Chief Safety Officer, said: “If you have been prescribed Sertraline 100mg tablets and have received batch number V2500425, please check the carton contains the right medication. You can find the batch number and expiry date printed on the side of the outer packaging.”
READ MORE: Anyone taking Metformin given warning over B12 side effect – but potential benefits
“If the blister strips inside the carton are labelled Citalopram 40mg, please contact your pharmacy as soon as possible. If they are labelled Sertraline 100mg, no further action is needed.
“Patients who have accidentally taken citalopram instead of – or as well as – sertraline, may experience some heightened serotonergic side effects. These can include nausea, headache, sleep changes, and mild anxiety.”
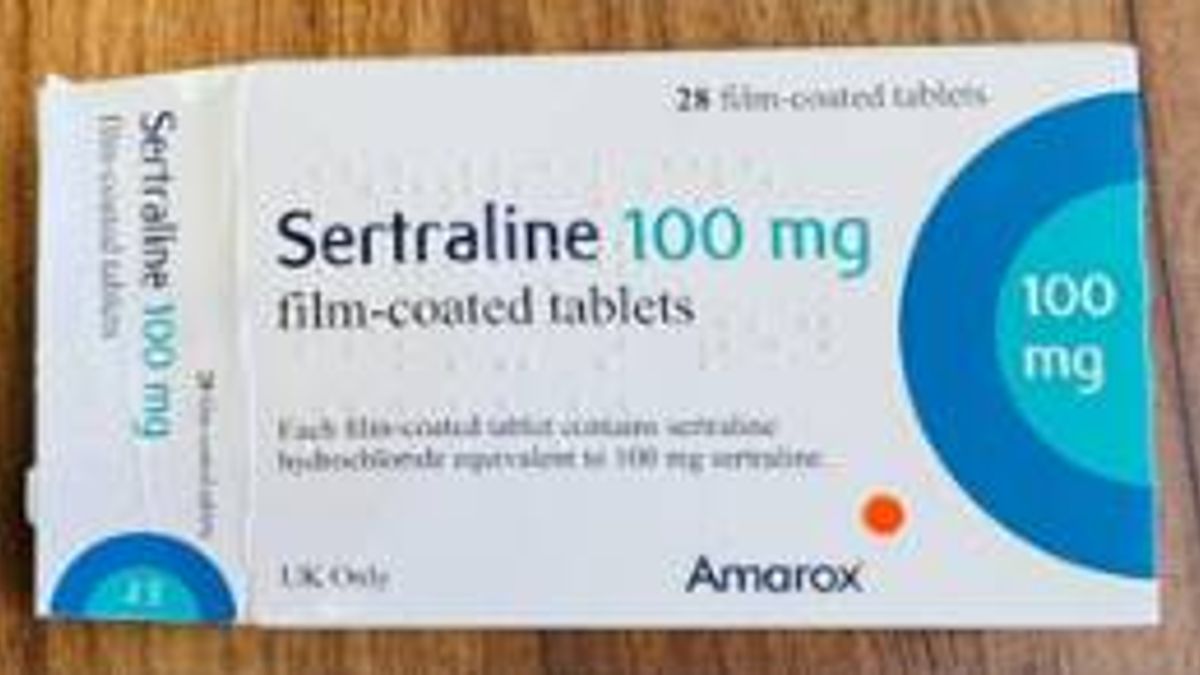
Sertraline 100 mg film-coated tablets(Image: MHRA)
Pharmacists and other healthcare professionals involved in dispensing who identify any patients with this product should get in touch with the patients’ GP, or the clinician responsible for their care, to discuss a treatment review and whether a fresh prescription is required for continued resupply.
Some patients may require monitoring by their doctor or another healthcare professional, particularly those who are over 65 or under 18, have cardiac or liver conditions, or have been advised that their body metabolises certain medicines differently.
The MHRA is urging anyone prescribed Sertraline to verify the batch number and expiry date printed on the side of their medication’s outer packaging. The regulator confirmed that the total batch size amounts to 81,872 packs. Boxes of 100mg film-coated sertraline tablets with batch number V2500425 and an expiry date of May 2028 have potentially been affected. The batch number and expiry date can be found printed on the cardboard carton provided by your pharmacist or medication supplier.
The affected batch was first distributed on 28 November 2025, meaning some boxes may still be unopened. A further indication that your prescription is affected is the presence of a strip of citalopram 40mg inside the box.
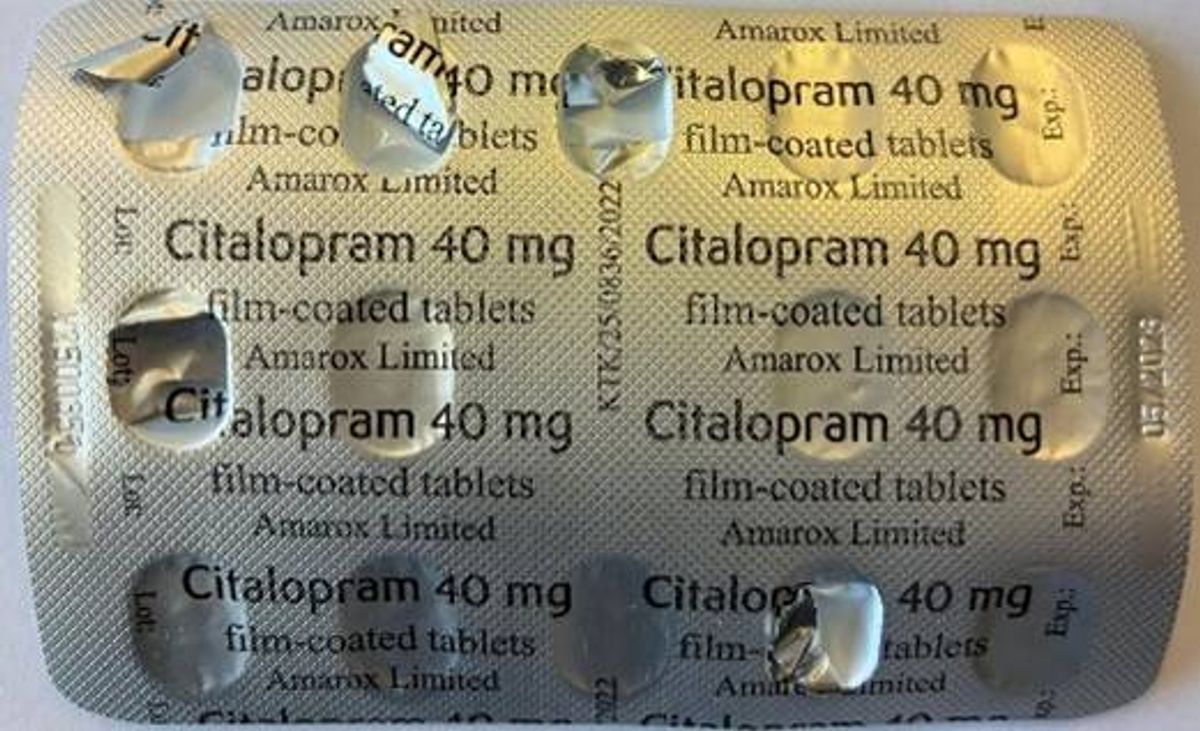
Citalopram 40 mg film-coated tablets – Batch Number V2500351 Incorrect blister strip(Image: MHRA)
This blister pack will have ‘citalopram 40mg’ clearly printed on the foil. Anyone discovering rogue strips of the drug has been urged to get in touch with their pharmacy straight away.
Pharmacists and other healthcare professionals involved in dispensing the antidepressant have also been advised to reach out to any patients who may have received the incorrect medication and request its return.
GPs and clinicians should then be notified of the mix-up to discuss a treatment review and whether a fresh prescription may be needed for continued supply. The MHRA warned that patients aged over 65 or under 18, along with those with heart or liver conditions, need to exercise particular caution.
What are the warning signs of serotonin syndrome? Those taking sertraline are being urged to ‘seek medical help immediately’ if they notice a rapid heartbeat, nausea, headaches or disrupted sleep patterns. These symptoms could indicate a potentially life-threatening condition known as serotonin syndrome – triggered by combining or switching between two different types of antidepressant.
Official NHS guidance confirms that serotonin syndrome symptoms can vary from mild to severe and require urgent medical attention. Physical warning signs can include hypertension – commonly known as high blood pressure – along with tachycardia, where the heart rate exceeds 100 beats per minute, and hyperthermia, which causes body temperature to soar to around 40C.
Further warning signs include dry eyes, unusually active bowel sounds, excessive sweating, tremors and clonus – involuntary, rhythmic muscle contractions. Patients may also suffer from muscle and joint stiffness, as well as hyperreflexia, a condition where reflexes become abnormally exaggerated.
For more information click here.
